Exoskeleton Research: Myoelectric orthosis for rehab of severe chronic arm motor deficits
A randomized controlled study investigating the efficacy of a myoelectric device combined with motor-learning therapy to improve arm function in chronic (>6 months) stroke survivors.

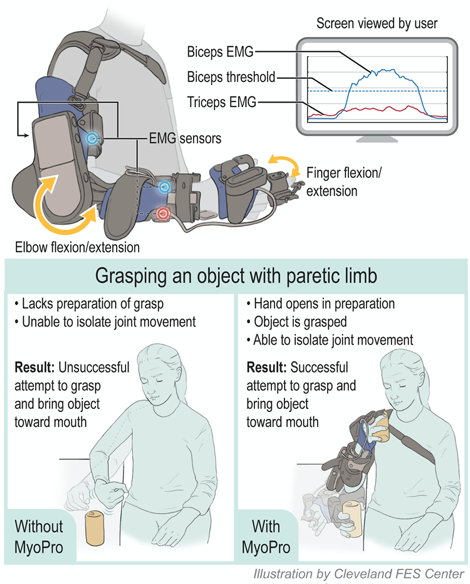
About this study
This study will evaluate the effects of combining motor learning-based therapy with use of the MyoPro, a wearable exoskeletal myoelectrically controlled orthotic device. MyoPro uses electromyographic (EMG) signals from the weak muscles to assist movement of the user’s affected arm. The primary objective of this randomized controlled trial is to study the efficacy of using MyoPro in motor learning-based therapy for individuals with chronic stroke (>6 months post) with severe upper limb motor deficits compared with a similar dose of motor learning-based therapy alone.
Study Information
Study participants are randomized into one of 2 groups: motor-learning only, or motor-learning plus MyoPro. All participants receive 9 weeks of in-clinic therapy (2 days/week) followed by 9 weeks of home practice with weekly check-in sessions with a physical therapist. Participants randomized to the motor-learning plus MyoPro group will receive a custom-fit MyoPro to use for the study duration. All participants will be assigned a personalized home exercise program. Measurements of the participants’ arm function are recorded before, during, and after active study participation. Changes in brain function will be evaluated for both groups to assess what additional benefit the MyoPro may provide in recovery of arm function.
ClinicalTrials.gov link: https://clinicaltrials.gov/study/NCT05296408
Principal Investigator: Svetlana Pundik, MD, MSc
Program Contact: Jessica McCabe, DPT
Contact Number: (216) 791-3800 ext. 63830
Contact Email: Jessica.McCabe@va.gov
Contact Request
Researchers rely on individuals to serve as volunteers for program studies. Each study is designed to answer questions about a specific medical aspect or the effectiveness of a particular treatment. Through the commitment of research volunteers, knowledge gained and communicated to other medical professionals ultimately benefits the community.
If you would like more information about becoming a research volunteer please submit the information below.
