Combining Non-invasive Brain Stimulation with Contralaterally Controlled FES for Upper Limb Rehabilitation after Severe Stroke
This study will analyze and compare different applications of repetitive transcranial magnetic stimulation plus contralaterally controlled FES to as post-stroke therapy programs.
This study is the first clinical trial of non-invasive brain stimulation (repetitive transcranial magnetic stimulation or rTMS) delivered to excite the undamaged hemisphere in stroke. Therefore, this study will determine whether the positive results obtained in the team’s short-term pilot study can be made to last longer and produce functional benefits in severe patients with the application of brain stimulation in combination with long-term rehabilitation therapy.
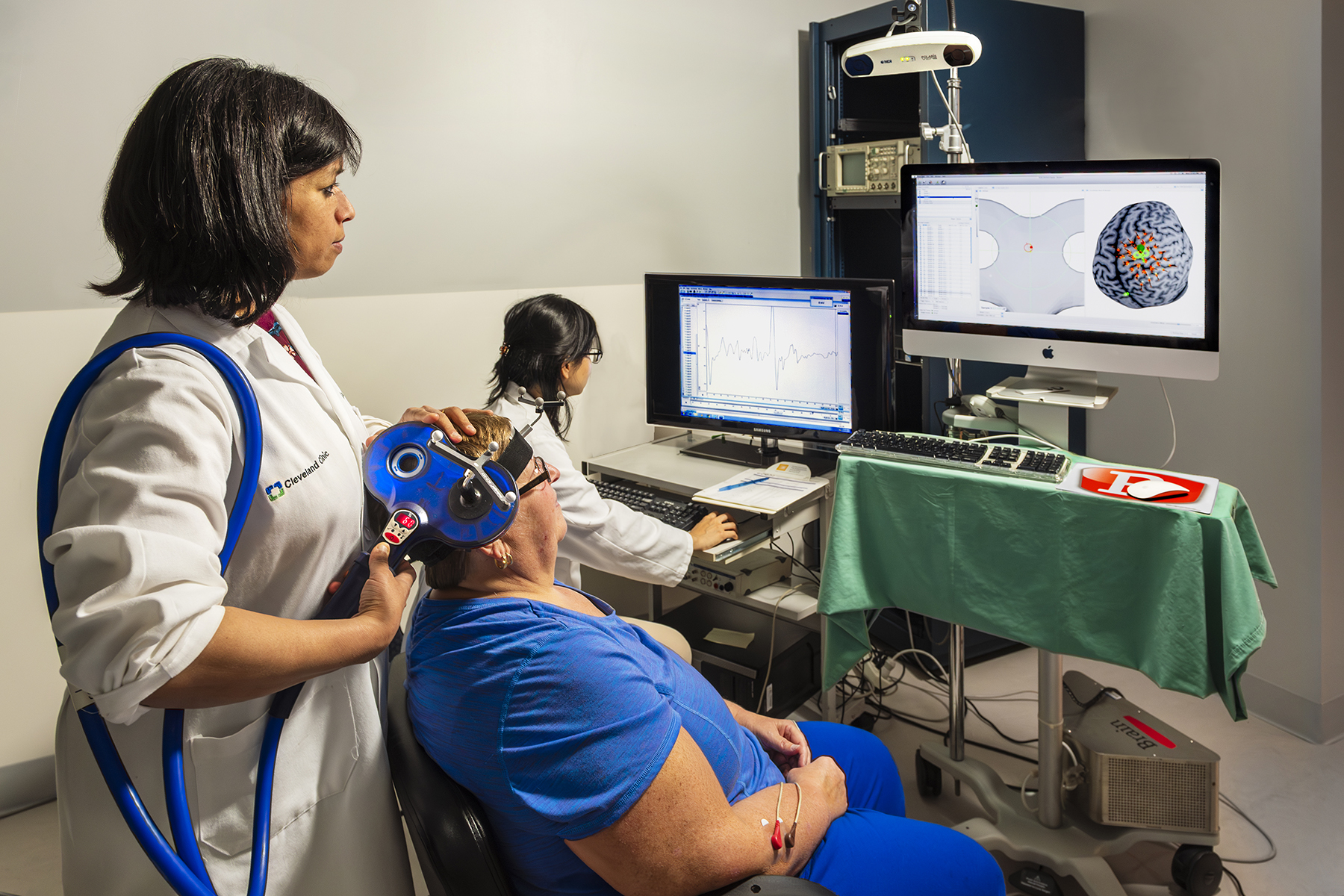
The rehabilitation therapy administered is called contralaterally controlled functional electrical stimulation (CCFES). The proposed study will determine whether the combination of rTMS facilitating the undamaged hemisphere with CCFES produces greater improvements in upper extremity function in severe participants who are >=6 months from stroke onset than the combination of rTMS facilitating the damaged hemisphere, and CCFES or the combination of sham rTMS and CCFES.
The secondary purposes of the study are to define which patients benefit most from the treatments, which may inform future device and treatment development and clinical translation, and to explore what distinct effects the three treatments have on the brain. To accomplish these purposes, the team is conducting a clinical trial that enrolls severe stroke patients.
Detailed study information is available at https://clinicaltrials.gov/study/NCT03870672.
For More Information
Contact Request
If you would like more information about becoming a research volunteer please submit the information below.