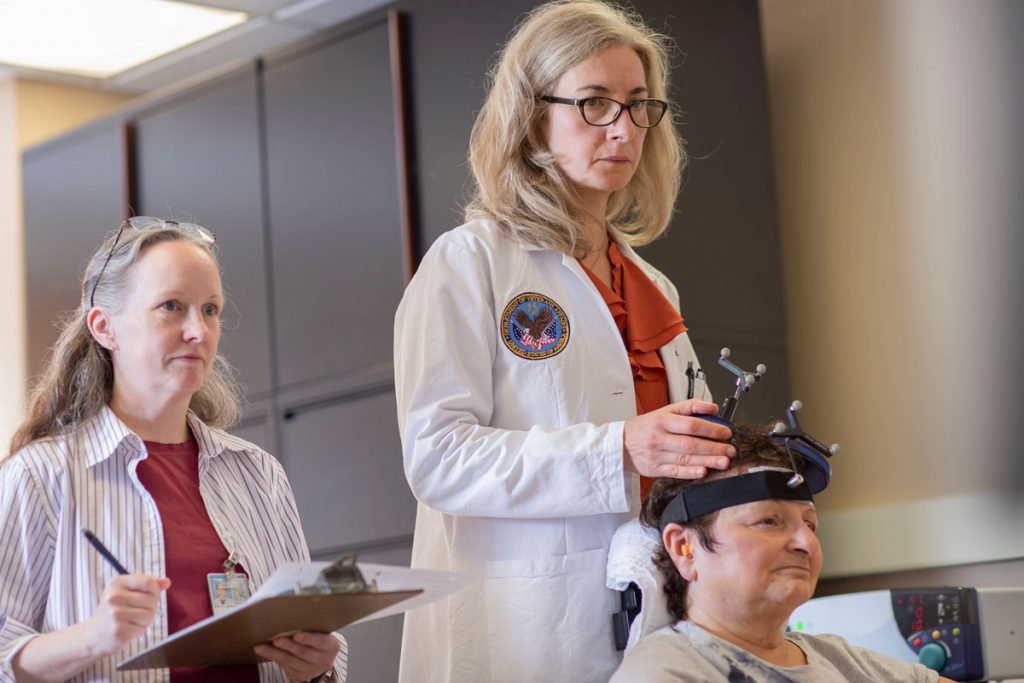
The therapy uses noninvasive brain stimulation called transcranial direct current stimulation (tDCS) in hopes of generating new pathways for brain neurons, which would allow stroke survivors to regain partial or full use of limbs. tDCS sends a low current between electrodes placed on the surface of the scalp.
Put in everyday terms, FES investigators are researching ways to use electricity to help stroke patients walk and use their hands again, a monumental step in recovery.
Pundik is one of several FES investigators focusing on neuroplasticity, the brain’s ability to change, grow and reorganize neural pathways and make new connections, in this case after stroke. The FES Center advances and propels the research of scientists, engineers and clinicians in the use of Functional Electric Stimulation (FES) to improve the life of those with neurological or other musculoskeletal impairments.
FES fosters a collaborative effort to find solutions via a consortium of five nationally recognized institutions: the Louis Stokes Cleveland VA Medical Center, Case Western Reserve University, MetroHealth Medical Center, University Hospitals and the Cleveland Clinic Neurological Institute.
By using neurostimulation and neuromodulation research and applications, the Cleveland FES vigorously seeks solutions as it fosters a culture of collaboration and support.
In the Gait program, Buckwald spends 15 minutes on a treadmill wearing devices on his head. Because this is a randomized control trial, the device may or may not be sending mild impulses into his brain.
As he walks the treadmill, virtual reality presents him a wooded pathway on a large video screen in front of him, with his footsteps appearing on the path.
Objects are placed on the right side of the path, forcing Buckwald to lift his right leg and even “hold” it over the object. That action prompts him to rely on his stroke-affected left leg, which allows his nerves to regain function, perhaps in a manner different from the way they functioned before the stroke.
“The overall hope is that the research will lead to a better understanding of how people recover after stroke, and to develop better ways of helping people recover,” says Pundik, a stroke neurologist, the Medical Director of the Stroke Program at the Louis Stokes Cleveland VA Medical Center, Director of the Brain Plasticity and NeuroRecovery Lab at FES Center and an Associate Professor of Neurology at CWRU School of Medicine.
The treadmill, which is just one part of Buckwald’s therapy regime, includes 34 “obstacles” and can move at varying speeds and inclines. Fourteen cameras measure precise details of the use of the impaired foot.
“Something is helping,” says Buckwald, who stays as active as he can, including cutting his lawn. “I know a lot of stuff is helping. The physical therapy is helping. The exercises are helping.”
Pundik’s research involves not only tDCS but also Transcranial Magnetic Stimulation (TMS) which uses a magnetic field to generate electrical currents in the brain.
Pundik concedes the challenges in helping stroke survivors are significant. As FES investigator Ahlam Salameh, PhD says, “Stroke is sudden and life changing, and not a lot of people handle it well.”
“We’re definitely on the cutting edge of where the electrical stimulation field is right now,” she says. “There are so many different options of how stimulation is applied, and different options for what the stimulation is paired with. Literally thousands of different options.”

The work takes time, and demands patience. While Parkinson’s and depression have a very consistent brain pathology, stroke injury is unique to each individual.
“So, it’s hard to find one treatment that fits everybody,” she says, “though we’re certainly trying to figure that out.”
A Hand in Daily Life
Jayme Knutson, PhD, another FES investigator focused on brain plasticity, uses CCFES in his work at Cleveland’s MetroHealth Medical Center. His focus is developing new therapies for stroke survivors who have lost hand function on one side of the body.
Knutson and his team put a glove on the good hand. The glove has sensors that can detect when the hand opens. That glove is connected to a stimulator on the affected arm.
“When they open the gloved hand, electrical stimulation opens the affected hand, and they can then practice using the affected hand to do tasks,” Knutson says.

“We still have a lot to learn about what’s happening in the brain, but it seems to respond to that,” Knutson says. “CCFES therapy seems to be sending signals back to the brain that are rewarding its attempt to open the hand.”
Neuroplasticity at work: over time and with therapy and repetition, the work coaxes the brain to change the way it sends signals to the impaired hand.
“We’ve done a number of studies in many stroke survivors, and we’ve seen positive outcomes from those studies,” Knutson says.
“A majority of people in the studies got an improvement in hand function that was significant, that made a difference for them, that helped them open their hand and use it more in their daily life.”

Knutson now is lead researcher on a multi-site study in Atlanta, New Jersey, Baltimore and Cleveland to see if other sites will see the same results.
He also developed a study with FES Investigator Michael Fu, PhD, that combines CCFES with custom-designed video games to challenge patients to achieve better control of finger opening.
“When therapists see the CCFES device on a video or at a presentation, they immediately want it,” Knutson says.
Time to Remember
Salameh’s work uses Near Infrared Spectroscopy and Imaging (NIRS), which can measure brain oxygenation during activity or rest.
Salameh explains that activities require energy, and energy means bringing blood to active tissue. Seeing that blood activity in the brain through infrared sensors gives researchers a picture of working pathways.
After stroke, the brain reverts to a developing-like state, she says. Repeated activity can re-teach the brain.
“It’s not building new neurons, just re-directing how the neurons connect with each other,” she says. “Making new networks, and new communications between neurons.”
In her work with research participants who have lost hand or arm function, Salameh uses a myoelectrically controlled device to support finger movement when even slight electrical activity is detected.
Therapy involves 18 sessions over a six-week period, 90 minutes per session. “You really have to repeat, repeat, repeat,” she says.
Salameh relayed a story of a participant who progressed from difficulty opening her hand to being able to grasp and move objects between tables.
“Oh, I screamed,” she says.
“It’s really nice because you see the individual as excited as when they took their first steps as a child all over again.”



